-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

B Braun 3175-11 - 2G Cefotetan for Injection and Dextrose Injection, Cephalosporins in the DUPLEX Container, 24 EA/CS
2G Cefotetan for Injection and Dextrose Injection, Cephalosporins in the DUPLEX Container
The DUPLEX two-compartment flexible IV container stores pre-measured drug and diluent doses separately until administration, while:
Being easy to use and convenient.
- Easy bedside activation
- No thawing or mixing procedures
- Simply fold and squeeze the bag to mix the pharmaceuticals and diluents just prior to administration
- Compatible with automated dispensing systems
- The only ready-to-use IV container with a portfolio covering four generations of cephalosporin products
- Simply label and dispense
- Automated charting via barcoding
- Store at room temperature
Enables compliance while protecting patients, providers and the environment.
- Prevents contamination and protects potency - closed system
- Helps reduce medication errors - no mixing required
- Protects medication efficacy - eliminates possibility of delivering diluents without drug
- Barcoding references final admixture
- Not manufactured with PVC, DEHP or natural rubber - removes potential for release of dioxins during disposal
- Economical and reduces drug waste
- Protects patients and healthcare workers from exposure to airborne cephalosporins
What is a DUPLEX Container?
DUPLEX is a two-compartment flexible plastic IV container that stores pre-measured drug and diluent doses separately until administration. DUPLEX is a ready-to-use, two-compartment container that keeps pre-measured medication and diluent separate until youre ready to mix and administer.DUPLEX containers are not made with natural rubber latex, DEHP or PVC.
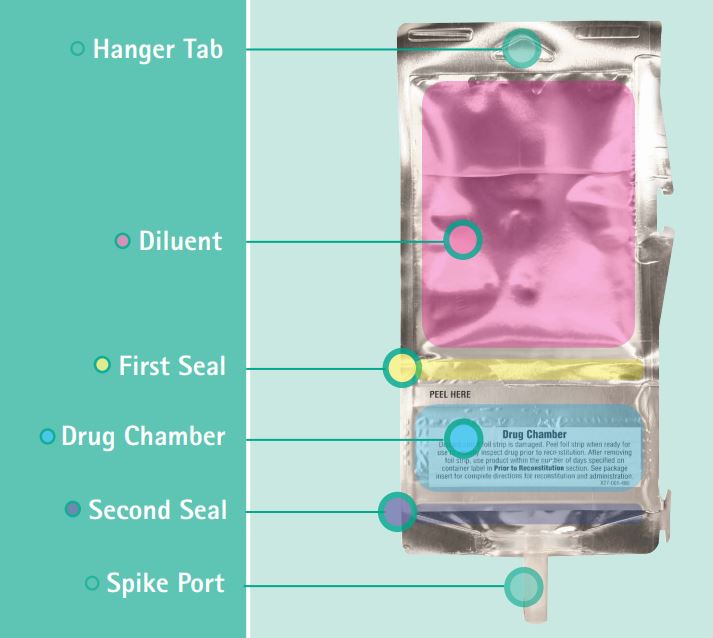
Benefits of DUPLEX Container for Facility Staff
- Simply fold and squeeze container to mix
- Saves time and labor for pharmacy and nursing
- Speeds dispensing, no vial connections or messy thawing
- Convenient - store at room temperature
- Automated charting via barcoding
- Economical - reduces drug lost to waste
- Compatible with automated dispensing systems
- All products offer 17 or more months expiration dating
- Meets Joint Commission and USP <797> guidelines
- Available in "most ready-to-use" form
Dramatically Reduces Potential for Medication Errors
- Protects medication efficacy - cannot deliver diluent without drug
- Prevents contamination and protects potency - closed system
- Reduces medication errors - no vial mixing required
- Protects patients and healthcare workers from exposure to airborne cephalosporins or carbapenemes
- Barcoding references final admixture, lot number and expiration date
- Not made with natural rubber latex, DEHP or PVC
Easy to Learn and Easy to Use
- Easy bedside activation: Simply break the quick release seal and squeeze to mix drug and diluent just prior to administration.
- Compatible with automated dispensing systems
- Simply label and dispense
- No vials to connect
- No thawing procedures
- Barcoding references final admixture, lot number and expiration date

DUPLEX Container Directions for Use
 | Label and Inspect:
NOTE: Product does NOT require refrigeration prior to activation. Color changes do not affect potency of drug. Reconstitute:
Administer:
Precautions:
|
Storage and Shipping
| Total Shelf Life (Months) | 018 |
|---|---|
Ordering Unit | Case |
Units per Case | 24 |
Dimensions per Case | 12.437 x 6.125 x 6.125 inches |
Shipping Weight per Case | 4.233 lb |


