-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Baxter 00641-6082-25 - PHENERGAN 25 MG AMP 25X1 ML, 25/BX
Phenergan Injection (Promethazine HCI Inj., USP), 25 mg/mL, 25 x 1 mL Ampuls, NDC 0641-6082-25
PHENERGAN Injection (promethazine hydrochloride injection, USP), is a sterile, pyrogen-free solution for deep intramuscular or intravenous administration. Promethazine hydrochloride (10H-Phenothiazine-10-ethanamine,N,N,a-trimethyl-, monohydrochloride, ()-) is a racemic compound and has the following structural formula:
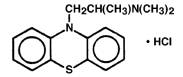
C17H21ClN2S MW 320.88
Each mL contains promethazine hydrochloride, either 25 mg or 50 mg, edetate disodium 0.1 mg, calcium chloride 0.04 mg, sodium metabisulfite 0.25 mg and phenol 5 mg in Water for Injection. pH 4.0 to 5.5; buffered with acetic acid-sodium acetate.
PHENERGAN Injection (promethazine hydrochloride injection, USP) is a clear, colorless solution. The product is light sensitive. It should be inspected before use and discarded if either color or particulate is observed.
Clinical Pharmacology
Promethazine hydrochloride is a phenothiazine derivative which possesses antihistaminic, sedative, antimotion-sickness, antiemetic, and anticholinergic effects. Promethazine is a competitive H1 receptor antagonist, but does not block the release of histamine. Structural differences from the neuroleptic phenothiazines result in its relative lack (1/10 that of chlorpromazine) of dopamine antagonist properties. Clinical effects are generally apparent within 5 minutes of an intravenous injection and within 20 minutes of an intramuscular injection. Duration of action is four to six hours, although effects may persist up to 12 hours. Promethazine hydrochloride is metabolized in the liver, with the sulfoxides of promethazine and N-desmethylpromethazine being the predominant metabolites appearing in the urine. Following intravenous administration in healthy volunteers, the plasma half-life for promethazine has been reported to range from 9 to 16 hours. The mean plasma half-life for promethazine after intramuscular administration in healthy volunteers has been reported to be 9.8 3.4 hours.
Indications and Usage
PHENERGAN Injection is indicated for the following conditions:
- Amelioration of allergic reactions to blood or plasma.
- In anaphylaxis as an adjunct to epinephrine and other standard measures after the acute symptoms have been controlled.
- For other uncomplicated allergic conditions of the immediate type when oral therapy is impossible or contraindicated.
- For sedation and relief of apprehension and to produce light sleep from which the patient can be easily aroused.
- Active treatment of motion sickness.
- Prevention and control of nausea and vomiting associated with certain types of anesthesia and surgery.
- As an adjunct to analgesics for the control of postoperative pain.
- Preoperative, postoperative, and obstetric (during labor) sedation.
- Intravenously in special surgical situations, such as repeated bronchoscopy, ophthalmic surgery, and poor-risk patients, with reduced amounts of meperidine or other narcotic analgesic as an adjunct to anesthesia and analgesia.
Contraindications
Children Less Than 2 Years of Age
PHENERGAN Injection is contraindicated for use in pediatric patients less than two years of age due to the risk of respiratory depression (see WARNINGS - Respiratory Depression).
Comatose State
PHENERGAN Injection is contraindicated in comatose states.
Intra-Arterial Injection
Under no circumstances should PHENERGAN Injection be given by intra-arterial injection due to the likelihood of severe arteriospasm and the possibility of resultant gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene).
Subcutaneous Injection
PHENERGAN Injection should not be given by the subcutaneous route because evidence of chemical irritation has been noted, and necrotic lesions have resulted following subcutaneous injection. The preferred parenteral route of administration is by deep intramuscular injection.
Idiosyncratic Reaction or Hypersensitivity
PHENERGAN Injection is contraindicated in patients who have demonstrated an idiosyncratic reaction or hypersensitivity to promethazine or other phenothiazines.
Warnings
Respiratory Depression
Pediatrics
PHENERGAN Injection should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression. Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age. A wide range of weight-based doses of PHENERGAN Injection have resulted in respiratory depression in these patients.
Caution should be exercised when administering PHENERGAN Injection to pediatric patients 2 years of age and older. It is recommended that the lowest effective dose of PHENERGAN Injection be used in pediatric patients 2 years of age and older. Avoid concomitant administration of other drugs with respiratory depressant effects because of an association with respiratory depression, and sometimes death, in pediatric patients.
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse event reports include burning, pain, erythema, swelling, sensory loss, palsies, paralysis, severe spasm of distal vessels, thrombophlebitis, venous thrombosis, phlebitis, abscesses, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required.
Because of the risks of intravenous injection, the preferred route of administration of PHENERGAN Injection is deep intramuscular injection (see DOSAGE AND ADMINISTRATION). Subcutaneous injection is contraindicated. Due to the close proximity of arteries and veins in the areas most commonly used for intravenous injection, extreme care should be exercised to avoid perivascular extravasation or unintentional intra-arterial injection as pain, severe chemical irritation, severe spasm of distal vessels, and resultant gangrene requiring amputation are likely under such circumstances. Aspiration of dark blood does not preclude intra-arterial needle placement because blood is discolored upon contact with PHENERGAN Injection. Use of syringes with rigid plungers or of small-bore needles might obscure typical arterial backflow if this is relied upon alone.
In the event that a patient complains of pain during intravenous injection of Phenergan Injection, the injection should be stopped immediately to evaluate for possible arterial injection or perivascular extravasation.
There is no proven successful management of unintentional intra-arterial injection or perivascular extravasation after it occurs. Sympathetic block and heparinization have been employed during the acute management of unintentional intra-arterial injection, because of the results of animal experiments with other known arteriolar irritants.
CNS Depression
PHENERGAN Injection may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. The impairment may be amplified by concomitant use of other central-nervous-system depressants such as alcohol, sedative/hypnotics (including barbiturates), general anesthetics, narcotics, narcotic analgesics, tricyclic antidepressants, and tranquilizers; therefore such agents should either be eliminated or given in reduced dosage in the presence of promethazine hydrochloride (see PRECAUTIONS - Information for Patients and Drug Interactions).
Lower Seizure Threshold
PHENERGAN Injection may lower seizure threshold and should be used with caution in persons with seizure disorders or in persons who are using concomitant medications, such as narcotics or local anesthetics, which may also affect seizure threshold.
Bone-Marrow Depression
PHENERGAN Injection should be used with caution in patients with bone-marrow depression. Leukopenia and agranulocytosis have been reported, usually when promethazine hydrochloride has been used in association with other known marrow-toxic agents.
Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with promethazine hydrochloride alone or in combination with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include 1) immediate discontinuation of promethazine hydrochloride, antipsychotic drugs, if any, and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
Since recurrences of NMS have been reported with phenothiazines, the reintroduction of promethazine hydrochloride should be carefully considered.
Sulfite Sensitivity
PHENERGAN Injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions, including anaphylactic symptoms and life-threatening or less severe asthma episodes, in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Visual Inspection
This product is light sensitive and should be inspected before use and discarded if either color or particulate is observed.
Cholestatic Jaundice
Administration of promethazine has been associated with reported cholestatic jaundice.
Precautions
General
Drugs having anticholinergic properties should be used with caution in patients with narrow-angle glaucoma, prostatic hypertrophy, stenosing peptic ulcer, pyloroduodenal obstruction, and bladder-neck obstruction.
PHENERGAN Injection should be used cautiously in persons with cardiovascular disease or impairment of liver function.
Pediatric Use
PHENERGAN Injection is contraindicated for use in pediatric patients less than 2 years of age, because of the potential for fatal respiratory depression. PHENERGAN Injection should be used with caution in pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression).
Antiemetics are not recommended for treatment of uncomplicated vomiting in pediatric patients, and their use should be limited to prolonged vomiting of known etiology. The extrapyramidal symptoms which can occur secondary to PHENERGAN Injection administration may be confused with the CNS signs of undiagnosed primary disease, e.g. encephalopathy or Reye's syndrome. The use of PHENERGAN Injection should be avoided in pediatric patients whose signs and symptoms may suggest Reye's syndrome or other hepatic diseases.
Excessively large dosages of antihistamines, including PHENERGAN Injection, in pediatric patients may cause sudden death (see OVERDOSAGE). Hallucinations and convulsions have occurred with therapeutic doses and overdoses of PHENERGAN Injection in pediatric patients. In pediatric patients who are acutely ill associated with dehydration, there is an increased susceptibility to dystonias with the use of PHENERGAN Injection.
Adverse Reactions
Respiratory Depression
PHENERGAN Injection is contraindicated in pediatric patients less than 2 years of age, because of the potential for fatal respiratory depression. PHENERGAN Injection should be used with caution in pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression).
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse reactions include burning, pain, erythema, swelling, sensory loss, palsies, paralysis, severe spasm of distal vessels, thrombophlebitis, venous thrombosis, phlebitis, abscesses, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required (see WARNINGS - Severe Tissue Injury, Including Gangrene; and DOSAGE AND ADMINISTRATION).
Central Nervous System
Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness, confusion, disorientation, and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Overdosage
Signs and symptoms of overdosage range from mild depression of the central nervous system and cardiovascular system to profound hypotension, respiratory depression, unconsciousness and sudden death. Other reported reactions include hyperreflexia, hypertonia, ataxia, athetosis, and extensor-plantar reflexes (Babinski reflex).
Stimulation may be evident, especially in pediatric patients and geriatric patients. Convulsions may rarely occur. A paradoxical-type reaction has been reported in pediatric patients receiving single doses of 75 mg to 125 mg orally, characterized by hyperexcitability and nightmares.
Atropine-like signs and symptoms-dry mouth; fixed, dilated pupils; flushing; etc., as well as gastrointestinal symptoms, may occur.
Treatment
Treatment of overdosage is essentially symptomatic and supportive. Only in cases of extreme overdosage or individual sensitivity do vital signs, including respiration, pulse, blood pressure, temperature, and EKG, need to be monitored. Attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. Diazepam may be used to control convulsions. Acidosis and electrolyte losses should be corrected. Note that any depressant effects of PHENERGAN Injection are not reversed by naloxone.
Avoid analeptics, which may cause convulsions. The treatment of choice for resulting hypotension is administration of intravenous fluids, accompanied by repositioning if indicated. In the event that vasopressors are considered for the management of severe hypotension which does not respond to intravenous fluids and repositioning, the administration of norepinephrine or phenylephrine should be considered. EPINEPHRINE SHOULD NOT BE USED, since its use in a patient with partial adrenergic blockade may further lower the blood pressure. Extrapyramidal reactions may be treated with anticholinergic antiparkinson agents, diphenhydramine, or barbiturates. Oxygen may also be administered. Limited experience with dialysis indicates that it is not helpful.
Dosage and Administration
Important Notes on Administration
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration (see WARNINGS - Severe Tissue Injury, Including Gangrene).
- The preferred parenteral route of administration for PHENERGAN Injection is by deep intramuscular injection.
- Under no circumstances should PHENERGAN Injection be given by intra-arterial injection due to the likelihood of severe arteriospasm and the possibility of resultant gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene).
- Subcutaneous injection is contraindicated as it may result in tissue necrosis.
- When administered intravenously, PHENERGAN Injection should be given in a concentration no greater than 25 mg per mL and at a rate not to exceed 25 mg per minute. It is preferable to inject through the tubing of an intravenous infusion set that is known to be functioning satisfactorily.
- In the event that a patient complains of pain during intravenous injection of PHENERGAN Injection, the injection should be stopped immediately to evaluate for possible arterial injection or perivascular extravasation.
Do not use PHENERGAN Injection if solution has developed color or contains precipitate.
To avoid the possibility of physical and/or chemical incompatibility, consult specialized literature before diluting with any injectable solution or combining with any other medication. Do not use if there is a precipitate or any sign of incompatibility.
Warnings
Respiratory Depression - Pediatrics
PHENERGAN Injection should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression. Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age. Caution should be exercised when administering PHENERGAN Injection to pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression).
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse reactions include burning, pain, thrombophlebitis, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required (see WARNINGS - Severe Tissue Injury, Including Gangrene).
Due to the risks of intravenous injection, the preferred route of administration of PHENERGAN Injection is deep intramuscular injection. Subcutaneous injection is contraindicated. See DOSAGE AND ADMINISTRATION for important notes on administration.

