-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Cook Medical G29953 - SET, BLOCKER, COHN, ENDO, C-AEBTS-9.0-65-SPH, EACH
Cohen Endobronchial Blocker Set
The Cohen Tip Deflecting Endobronchial Blocker consists of a blocker catheter and a multi-port airway adapter. A suction adapter is provided for an alternative method of lung deflation, if desired. The endobronchial blocker uses a tip-deflecting mechanism to allow the tip to be directed into the right or left mainstem bronchus. A small-diameter (pediatric) fiberoptic bronchoscope is used to visualize and aid in the placement of the endobronchial blocker. The lung may then be blocked by inflating the balloon within either the right or left mainstem bronchus.
The endobronchial blocker does not obstruct the working channel of the bronchoscope during placement. Refer to label for further information.
Included Components
- Arndt Multi-Port Adapter
- CPAP Adapter
- Syringe
- Suction adapter
Features and benefits
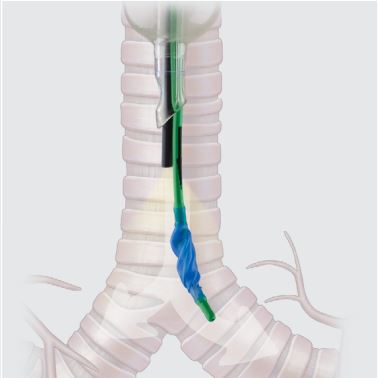 |
2. The high-traction silicone grip on the introducer shaft 3. The low-pressure, high-volume balloon creates 4. Distance markings increase visualisation by indicating 5. The suction adapter included with the set offers an |
Intended Use
The Cohen Tip Deflecting Endobronchial Blocker is intended for use to differentially intubate a patients bronchus in order to isolate the left or right lung for procedures that require one-lung ventilation. The product is intended for use by physicians trained and experienced in one-lung ventilation. Standard techniques for placement of endobronchial blockers should be employed.
Contraindications
- Airway or endotracheal tube diameter insufficient to allow passage of the Cohen Tip Deflecting Endobronchial Blocker.
- Fiberoptic equipment is unavailable. It is not possible to properly position the blocker without a fiberoptic bronchoscope.
Warnings
- Always advance the endobronchial blocker under direct fiberoptic vision.
- Do not advance the endobronchial blocker if resistance is encountered.
- If ventilation should become acutely difficult during endobronchial blockade, the balloon should be immediately deflated and the balloon position inspected.
- Caution is recommended when working near the hilum. The balloon position should be verified to prevent inadvertent balloon damage or movement. This is particularly recommended during right-sided procedures in which the balloon is positioned near the carina.
- As per all cases of one-lung ventilation, the use of pulse oximetry is mandatory when using this device.
- It may not be possible to adequately ventilate a patient during placement of the endobronchial blocker due to the presence of a bronchoscope and the endobronchial blocker in the endotracheal tube.
- An increased FiO2 for the ventilated lung is often needed to help maintain adequate arterial oxygen saturation.
- In some circumstances, an increased FiO2 for the ventilated lung may not be sufficient to maintain adequate arterial oxygen saturation, and other standard techniques routinely employed during one-lung ventilation may be necessary.
- Placement in the right mainstem bronchus may result in herniation of the balloon into the right upper lobe bronchus and thereby occlude it.
- During one-lung ventilation, the patient should be paralyzed to help prevent dislodgement of the balloon.
- If any resistance is met in removing the blocker after surgery, the blocker tip should be inspected fiberoptically to ensure integrity.
Precautions
- The endobronchial blocker balloon is a high-volume, low-pressure design. Excessive manipulation over a prolonged period may cause balloon rupture or deflation.
- This device should only be used by those familiar with the use of small diameter (pediatric) fiberoptic bronchoscopes and airway anatomy.
- Retract balloon prior to bronchial clamping during pneumonectomy procedures.
- Inflate balloon initially under direct vision to ensure correct position and placement.
- Ensure the balloon remains fully inflated during longer (>1 hour) procedures.
- For best results, initially, the blocker should be placed with the patient in a supine position, although the blocker can be easily and safely directed to the desired bronchus when the patient is in the lateral decubitus.
- To avoid damage to the small-diameter fiberoptic bronchoscope, standard lubricant may be used for the bronchoscope and the blocker.
- Reconfirm balloon position after positioning the patient for the procedure.
- Securely tape in place the endotracheal tube during use of the endobronchial blocker.
- During use of the endobronchial blocker, the capnograph waveform may become distorted.
- The blocker can be placed through a standard endotracheal tube. For optimal performance, the largest endotracheal tube is recommended.
- The smallest recommended size endotracheal tube for use with the 9.0 French blocker is 7.0 mm.
- Following insertion through the multi-port adapter, test inflate the balloon.
- If using supplied suction adapter during lung deflation procedure, ensure that you do not inadvertently attach it to the pilot balloon assembly. Suction adapter should only be attached to Luer lock connector.
- The potential effects of phthalates on pregnant/nursing women or children have not been fully characterized and there may be concern for reproductive and developmental effects.
Potential Adverse Events
The adverse events are those of lung isolation and one-lung ventilation:
- Hypoxia
- Hypoxemia
- Endobronchial irritation
- Tracheal injury
How Supplied
Supplied sterilized by ethylene oxide gas in peel-open packages. Intended for one-time use. Sterile if package is unopened or undamaged. Do not use the product if there is doubt as to whether the product is sterile. Store in a dark, dry, cool place. Avoid extended exposure to light. Upon removal from package, inspect the product to ensure no damage has occurred.

