-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Cook Medical G31455 - PATCH SURGISES PARAESOPHEGEAL, EACH
Biodesign Hiatal Hernia Graft Preshaped
The Biodesign Hiatal Hernia Graft is a multi-layered sheet of extracellular collagen matrix derived from porcine small intestinal submucosa. The Biodesign Hiatal Hernia Graft is implanted by a trained medical professional to support weakened soft tissue during surgery to repair hiatal hernias.
| Order Number | Reference Part Number | Size cm |
| G31455 | C-PHR-7X10-U | 7 x 10 |
Intended Use
The Biodesign Hiatal Hernia Graft is used for implantation to reinforce soft tissue where weakness exists, including repair of hiatal hernias. This graft is supplied sterile in peel-open packages and is intended for one-time use.
CAUTION: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Contraindications
This graft is derived from a porcine source and should not be used for patients with known sensitivity to porcine material.
Precautions
- This device is designed for single use only. Attempts to reprocess, resterilize, and/or reuse may lead to device failure and/or transmission of disease.
- Do not resterilize. Discard all open and unused portions.
- Graft is sterile if the package is dry, unopened and undamaged. Do not use if the package seal is broken.
- Discard graft if mishandling has caused possible damage or contamination, or if the graft is past its expiration date.
- Ensure that the graft is rehydrated prior to cutting or suturing. Insufficient hydration may cause device damage during laparoscopic delivery.
- Ensure that all layers of the graft are secured when suturing or stapling.
- Ensure that the graft does not touch the esophagus.
Potential Complications
The following complications are possible with the use of surgical graft materials.
- Infection
- Acute or Chronic inflammation (Initial application of surgical graft materials may be associated with transient, mild, localized inflammation.)
- Allergic reaction
- Adhesion
- Fistula formation
- Seroma formation
- Hematoma
- Recurrence of tissue defect
The following complications are associated with use of graft materials in hiatal hernia repair.
- Esophageal erosion
- Stricture formation
- Perforation
- Dysphagia
Suggested Instructions for Using The Hiatal Hernia Graft
Required Materials
- A sterile dish (kidney dish or other bowl)
- Sterile forceps
- Rehydration fluid: room temperature sterile saline or sterile lactated Ringers solution
NOTE: Handle sheets using aseptic technique, minimizing contact with latex gloves.
- Using aseptic technique, remove the graft inner pouch from its outer bag, and place the inner pouch in the sterile field.
- Open the inner pouch carefully, and aseptically remove the graft with the sterile forceps.
- Place the graft into the sterile dish in the sterile field. (Multiple grafts may be rehydrated simultaneously in the same dish.)
- Add to the dish at least 50 mL of the rehydration fluid for each graft.
- Rehydrate the graft in the rehydration fluid until the desired handling characteristics are achieved. A rehydration time of greater than 1 minute is not required.
- Prepare the graft site using standard surgical techniques.
NOTE: Surgical experience indicates that suturing or stapling the graft with close tissue approximation produces better outcomes. Maximum graft-tissue approximation is only achieved with crural closure - Using aseptic technique, trim the graft to fit the site, if necessary, providing a small allowance for overlap.
NOTE: An alternative method is to cut the graft prior to rehydration to fit the patients anatomy. If this method is selected, be sure to rehydrate the graft prior to suturing or stapling it into place. See step 5. - Using aseptic technique, transfer the graft to the graft site and suture or staple into place, avoiding excess tension.
NOTE: The 7X10-U graft has a larger hole in the corner to identify graft orientation. It is recommended that the larger hole should be placed at the patient upper left.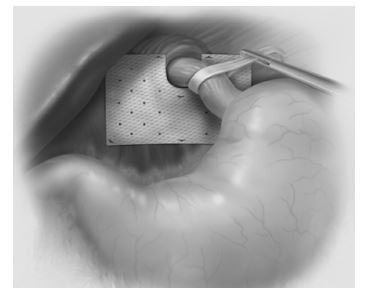
NOTE: The 7X10-AP graft should be oriented such that the wide arm reinforces the posterior space while the small arm reinforces the anterior space.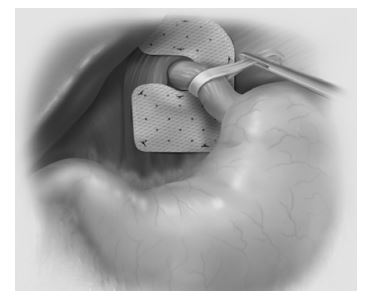
- Complete the standard surgical procedure.
- Discard any unused portions of the graft according to institutional guidelines for disposal of medical waste.
Sterilization
This graft has been sterilized with ethylene oxide.
Device Characteristics
| What MRI safety information does the labeling contain? | Labeling does not contain MRI Safety Information |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437): | No |
| Device labeled as "Not made with natural rubber latex": | No |
| For Single-Use: | Yes |
| Prescription Use (Rx): | Yes |
| Over the Counter (OTC): | No |
| Kit: | No |
| Combination Product: | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P): | No |

