-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Cook Medical G46045 - DEVICE, BIOPSY, BX, EACH

Cook Bx Biopsy Device
The Cook Bx Biopsy Device is designed to harvest multiple high-quality histological tissue specimens from the prostate or kidney. The Cook Bx is reusable, which makes it environmentally friendly and minimizes the quantity and cost of disposable waste. The device is designed for use with the Cook Biopsy Needle.
Set components may vary:
- Bx Core Tissue Biopsy Device
- Periprostatic injection needle (for transrectal local anesthetic injection)
- Needles
Features
- Easy specimen retrieval that doesnt require removing the needle from the instrument.
- Powerful dual-spring design for rapid-fire action.



- Easy actuation of trigger from front or rear of instrument for single-person operation.
- Automatic safety to prevent accidental firing.
- Small, lightweight design.
Intended Use
The Cook Bx Device is a reusable system for histological core biopsies. It has a throw (advancement) of 25mm and is used in conjunction with a single use Cook Bx Needle. The Cook Bx Device is used to obtain tissue core biopsy of the prostate using only the Cook Bx Needle. These instructions for the Cook Bx Device are not meant to define or suggest any medical or surgical technique. The individual practitioner is responsible for the proper procedure and techniques to be used with this device.
Contraindications
For use only for core biopsies of soft tissue as determined by a licensed physician. The device should be used by a physician familiar with the possible side effects, typical findings, limitations, indications and contraindications of core needle biopsy. Physician judgment is required when considering biopsy on patients with a bleeding disorder, or receiving anti-coagulant medications.
Device Characteristics
| What MRI safety information does the labeling contain? | MR Unsafe |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437): | No |
| Device labeled as "Not made with natural rubber latex": | No |
| For Single-Use: | Yes |
| Prescription Use (Rx): | Yes |
| Over the Counter (OTC): | No |
| Kit: | No |
| Combination Product: | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P): | No |
Precautions
- Not closing the lid may lead to malfunction of the device.
- Do not use device in presence of Magnetic Resonance Imaging (MRI).
Instructions for Using The Cook Bx Device And Needle
- Inspect the product package for damage. If undamaged, open the package using proper aseptic technique.
- Remove the needle from the pouch. Inspect the stylet for damage to the point, the cannula for damage to the cutting edge, and check for other imperfections that would prevent the proper operation of the needle assembly.
- Open the lid of the biopsy device, load needle set into the biopsy device, close lid, and remove protective sheath.
- Safety button must be set in the unlocked (right) position before cocking.
- Cock the actuator handle twice to activate the device for firing (this automatically sets the safety.)
- Release the safety by pushing the Safety Release Button located on the side of the device.
- Firing the device can be achieved by pushing in on either the front trigger button or the rear trigger button . This action will trigger the spring mechanism, propelling the needle forward for a core biopsy sample.
- The needle need not be removed from the biopsy device to retrieve the sample.
- After removing the needle from the patient, reset safety button and cock the actuator handle once to expose the biopsy notch located at the distal end of the needle. This action exposes the specimen for removal. Cocking the actuator handle a second time will activate the device for a second biopsy.
- The disposable needle set is for Single Patient Use Only. Do not attempt to clean or resterilize the needle set. After use, this product may be a potential biohazard. Handle in a manner that will prevent accidental puncture. Dispose in accordance with applicable laws and regulations.
Instructions for Cleaning The Cook Biopsy Device
- The Cook Bx Device may be cleaned in a mild detergent and lukewarm water solution. Never place the device in bleach or alcohol, or in any bleach, or alcohol solution.
- After washing completely, rinse the device in hot water.
- A high level of disinfection can be achieved with CIDEX 7 or ProCide NS: follow the manufacturers recommended procedure.
- Thoroughly dry the device to ensure proper function.
Autoclave Sterilization
Sterilizing by normal autoclaving may be employed. The device should first be cleaned and placed in appropriate packaging materials capable of maintaining sterility. Although cycle conditions and times will vary, typical conditions for autoclaving are 270? F (132?) for 15 to 25 minutes. To ensure effectiveness, sterilization processes should be properly validated and monitored with the proper biological controls. Sterilization operations should proceed according to the sterilizer manufacturers instructions.
NOTE: Do not sterilize by irradiation or gas plasma sterilization.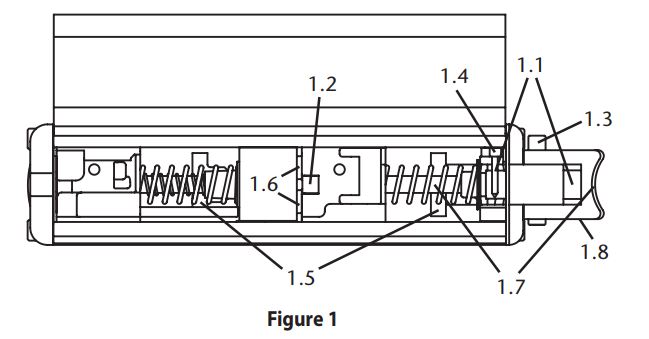
Lubrication Instructions
Washing detergents may eventually remove the lubricant originally applied. Re-lubrication at the following points may be required: (Refer to figure 1)
- 1.1 Trigger Button (must move freely)
Recommended Lube: B - 1.2 Top End of Actuator Pin
Recommended Lube: B - 1.3 Safety Button (must move freely; lube both sides)
Recommended Lube: B - 1.4 Lever Pivot
Recommended Lube: B - 1.5 Top Edge of Ramps on Latch
Recommended Lube: A - 1.6 Sides of Catch on Stylet Holder Block
Recommended Lube: B - 1.7 Cam Surfaces
Recommended Lube: A - 1.8 Link Pivots
Recommended Lube: B
Lubricant A:
- Permatex Superlube Synthetic Grease with Teflon
Lubricant B:
- Permatex Superlube Synthetic Oil with Teflon
Both Lubricants are U.S.D.A. approved for food grade applications and available locally in hardware and automotive stores. Use lubricants sparingly.
IMPORTANT: Lubrication on the sides of the carrier blocks will slow the action of the device.
How Supplied
Supplied non-sterile in peel-open packages. Reusable. Store in dark, dry cool place. Avoid extended exposure to light. Upon removable from package, inspect the product to ensure no damanage has occured Needles are sterile (ETO) if package is unopened or undamaged.

Cook Medical #G08789, SET, NEEDLE, QUICK-CORE, COAXIAL, BIOPSY, EACH
$203.30 EACH

Cook Medical #G27018, SET, NEEDLE QUICK-CORE COAXIAL BIOPSY, EACH
$203.28 EACH

Cook Medical #G31521, NEEDLE, ULTRA SOUND, ENDOSCOPIC, ECHOTIP, EACH
$570.88 EACH

Cook Medical #G08790, SET, QUICK-CORE, COAXIAL, BIOPSY, NEEDLE, EACH
$198.73 EACH

