-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Medline MSC2710 - DRESSING, TRNSPRNT, SURESITE123, 4"X10", 25 EA/BX, 4 BX/CS
Suresite 123 Transparent Film Dressing
Transparent film dressing with easy 1-2-3 delivery!
Transparent Film Dressings can be used to cover and protect catheter sites and wounds, to maintain a moist environment for wound healing or to facilitate autolytic debridement, as a secondary dressing, as a protective cover over at-risk skin, to secure devices to the skin and as a protective eye covering. Common applications include a variety of I.V. catheters and other percutaneous devices.
- Easy to apply transparent film
- Traditional moisture vapor transmission rate (MVTR)
- Conformable; won't stick to itself
- Microporous technology
- Permits continuous observation
- Variety of delivery systems
About Suresite
Transparent Film Dressings
- Traditional moisture vapor transmission rate (MVTR)
- Conformable
- Does not stick to itself
- Microporous technology
- Permits continuous observation of wound and line sites
- Variety of delivery systems
Indications
- Peripheral and central I.V. lines
- Partial-thickness wounds
- Full-thickness wounds (secondary dressing)
- Lacerations and abrasions
- To help prevent skin breakdown caused by friction to epidermis
Contraindications
- Contraindicated as a primary dressing on wounds with moderate to heavy drainage
Change Frequency
- Suresite may be left in place for up to 7 days
- Dressing change frequency will depend on amount of drainage
Recommended Uses
- Shallow
- No/minimal drainage
- Primary dressing
- Secondary dressing
Recommended Secondary Dressings
- Not Applicable
MVTR (Moisture Vapour Transmission Rate)
Moisture vapour transmission rate (MVTR) is the measurement of water vapour diffusion through a material.
Two laboratory test methods are commonly used to measure MVTR. The results of these two tests are often used to compare transparent dressings for I.V. use; however, they do not represent real life conditions, and numerous variables can impact the results. This raises the question of whether laboratory test data for MVTR can accurately predict dressing performance in clinical practice.

The inverted beaker test produces higher numbers with greater variability. These variances are seen within samples of the same dressing, as well as among different products. This inconsistency occurs because the films can stretch and swell due to the water pressure against the test dressing, increasing surface area measured. The MVTR values produces by the upright method are lower and more consistent among different products, and within samples of the same dressing. Due to the fact that the liquid does not come in contact with the film in this test method, stretch and swell are not factors in the results.
Aside from the test method chosen, many other variables can significantly affect MVTR values:
- Volume of liquid in the test beaker (generally 10-50 ml)
- Type of liquid medium (water, saline)
- Concentration of substances in the liquid (salt, proteins)
- Environmental conditions (temperature, humidity)
Research studies have been conducted to investigate the effect of MVTR on clinical outcomes for I.V. therapy. The results of these trials do not demonstrate a correlation between higher MVTR values and lower incidence of complications, including catheter-related bacteremia.
To date, there is no specific clinical evidence to suggest the optimal moisture vapour transmission rate. More important than MVTR in preventing I.V. infections are proper site preparation, sterile insertion technique, and strict adherence to protocols for I.V. site maintenance.
Specifications
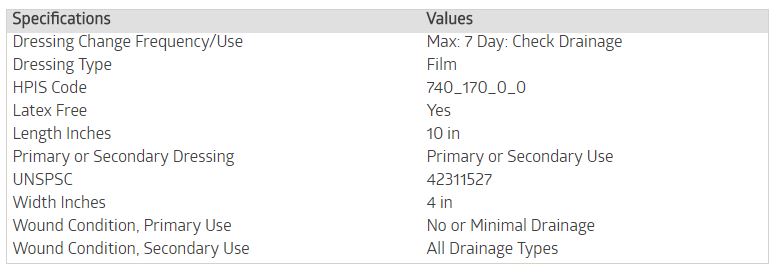

Medline #MSC2710, DRESSING, TRNSPRNT, SURESITE123, 4"X10", 25 EA/BX, 100 EA/CS
Call for Pricing

Medline #MSC2706, DRESSING, TRNSPRNT, SURESITE123, 6"X8", 25 EA/BX, 4 BX/CS
Call for Pricing

Medline #MSC2712, DRESSING, TRNSPRNT, SURESITE123, 8"X12", 25 EA/BX, 4 BX/CS
Call for Pricing

