-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Smith & Nephew 66027643 - DRESSING, FOAM, ALLEVYN, NON-ADHESIVE, 2"X2", 60 EA/CS, 6 BX/CS
ALLEVYN Ag Non-Adhesive - Advanced Foam Wound Dressings, 10cm x 10cm
From the beginning ALLEVYN was designed to respond to the human body. ALLEVYN Non-Adhesive was the first ALLEVYN variant, launched in 1987, and it combined patient comfort with the revolutionary trilayer fluid management system that enables moist wound healing.
ALLEVYN Non-Adhesive has stood the test of time and continues to be an essential for patients and clinicians all over the world.
- Unique triple action technology effectively manages fluid to create a moist wound healing environment
- Highly breathable top film helps to minimise the risk of maceration to the wound and periwound
- Non-adherent wound contact layer means the dressing is suitable for use on fragile and sensitive skin including epidermolysis bullosa wounds
- Soft and comfortable for the patient and conformable to challenging body areas
- Specific Heel "cup" dressing designed to conform to this awkward area
- Easy to apply and remove
- Provides an effective barrier function to exogenous bacteria as well as helping to prevent fluid and bacterial strikethrough
- Wide range of sizes that can be cut to suit different body contours
- Suitable for use under compression bandaging
- ALLEVYN Heel can contribute to a pressure relieving protocol when used prophylactically
- Shallow, granulating wounds
- Chronic and acute exudative wounds
- Full and partial thickness wounds such as pressure ulcers, leg ulcers and diabetic foot ulcers
- Infected wounds
- Malignant wounds
- Surgical wounds
- First and second degree burns
- Donor sites
- Fungating ulcers
- Oncological Wounds
- Epidermolysis bullosa wounds
ALLEVYN Non-Adhesive utilises a triple layer construction incorporating a highly absorbent hydrocellular foam between a breathable top film1 and a non-adherent wound contact layer to combine patient comfort with effective moisture control.
- Highly breathable top film
- helps to minimise the risk of maceration to the wound and peri-wound
- Non-adherent wound contact layer means the dressing is suitable for use on fragile and sensitive skin including epidermolysis bullosa wounds
- Soft and comfortable for the patient and conformable to challenging body areas
- Specific Heel "cup" dressing designed to conform to this awkward area
- Wide range of sizes that can be cut to suit different body contours
- Suitable for use under compression bandaging
- Easy to apply and remove
- Specific tracheostomy shape designed to provide absorption at tracheostomy and drainage sites
Technology
ALLEVYN Non-Adhesive utilises the unique triple action technology seen across the ALLEVYN range of dressings. By combining an absorbent hydrocellular pad with a non-adherent wound contact layer and breathable top film; the dressing manages fluid to maintain optimal moist wound healing conditions.
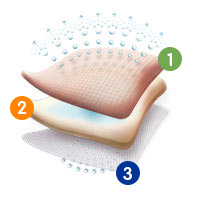
Breathable Top Film
The outer polyurethane top film is highly breathable, helps to prevent strikethrough and provides an effective barrier to bacteria.
Highly Absorbent Foam Core
The hydrocellular foam core is highly absorbent
Non-Adherent Wound Contact Layer
The wound contact layer will not stick to the wound and is suitable for fragile skin.
ALLEVYN Non-Adhesive is ideal for fragile and sensitive skin due to its non-adherent wound contact layer and soft and conformable construction. The specially designed Heel dressing fits closely to the heel area allowing even this most challenging of body parts to be dressed comfortably.

Indications
Shallow, granulating wounds, chronic and acute exudative wounds, full and partial thickness wounds such as pressure ulcers, leg ulcers and diabetic foot ulcers, infected wounds, malignant wounds, surgical wounds, first and second degree burns, donor sites, fungating ulcers, oncological wounds and epidermolysis bullosa wounds.
Application
Cleanse wound in accordance with local clinical protocols. Select an appropriate dressing size. Prepare and clean the skin surrounding the wound area and remove excess moisture. Any excess hair should be clipped to ensure close approximation to the wound. Remove ALLEVYN Non-Adhesive from the pack using a clean technique. Apply the white face to the wound ensuring good contact and secure with a dressing retention sheet (eg OPSITE Flexifix or OPSITE Flexifix Gentle, tape or bandage). When used under compression therapy, the dressing must be cut to the size of the wound. ALLEVYN Non-Adhesive can be cut to fit awkward areas. Always use a clean technique when cutting the dressing. If the dressing has been cut ensure any exposed foam areas are covered with an appropriate film dressing taking care not to cover the entire dressing.
Frequency of change
During the early stages of wound management ALLEVYN Non Adhesive should be inspected according to local clinical protocol. Where the product is used on infected wounds the infection should be inspected and treated as per local clinical protocol. Dressings can be left in place, undisturbed, for up to 7 days, or until exudate is visible and approaches 1.5cm from the edge of the dressing, whichever is sooner. If dressings are applied to the sacral area, they may be left in place for up to 5 days, however, due to the increased potential for contamination and infection in this area, increased monitoring of dressing adherence may be required as per local clinical protocols. The decision of when to change should be dependent upon clinical assessment and local protocols should also be taken into consideration.
Precautions
Do not use ALLEVYN Non-Adhesive with oxidising agents such as hypochlorite solutions (eg. EUSOL) or hydrogen peroxide, as these can break down the absorbent polyurethane component of the dressing. If reddening or sensitisation occur, discontinue use and consult a healthcare professional. ALLEVYN Heel must not be used for pressure relief independently of pressure relieving devices. ALLEVYN Non-Adhesive is a single use product. If used on more than one patient, cross-contamination or infection may occur. Opening the dressing pack compromises the sterile barrier therefore any unused dressing should not be retained for use at a later date.


Smith & Nephew #66020977, DRESSING, NON-ADHESIVE, ALLEVYN, 2"X2", 60 EA/CS
$661.07 per CASE

Smith & Nephew #66027643, DRESSING WOUND FOAM 2X2, 60 PER/CS
$185.23 per CASE

Smith & Nephew #66007630, DRESSING, FOAM, ALLEVYN, HEEL, 4.5"X5.5", 30 EA/CS, 6 BX/CS
$498.46 per CASE

Smith & Nephew #66800836, ALLEVYN, GB, 2"X5", 60 EA/CS, 6 BX/CS
$262.58 per CASE


